
Diagram 6. Nitrogen triiodide, dried compound.
3. Nitrogen triiodide and agar.
3.1 Formation, formula and chemical properties of NI3
The equation for the formation of NI3 from iodine and ammonia is
3I2 + NH3 -> NI3 + 3HI
Nitrogen triiodide is formed by the reaction between iodine solid and ammonia (0.880 S.G.). The solid is dark brown/black in colour. The solid chemical exists in one of two forms1.
Both of these are known to exist (there is XRD evidence for both as well as IR and VIS spectroscopy results).
When the NI3 is in the presence of ammonia solution, it is very stable for transportation and study. When the ammonical solution is removed, the amount of energy keeping the adduct together is too small, so the ammonia evaporates off, leaving the highly unstable solid.
The instability is caused by the large differences in atomic size. There is simply not enough room around the central nitrogen to keep all three iodine atoms stable (diagram 6 shows the structure of the unbound NI3. Diagram 7 shows ammonia.). The amount of energy required to detonate the dried chemical is a matter of Joules per mole, compared with the reaction output. The amount of energy given out on detonation is far greater than that required to form the compound.

Diagram 6. Nitrogen triiodide, dried compound.
The definition of a high yield explosive is that the amount of energy required to form the explosive is far less than that produced on explosion.
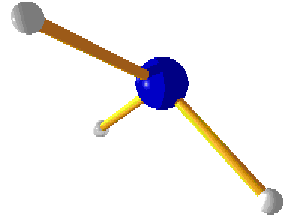
Diagram 7. Ammonia.
3.2 Preparation of the adduct
Traditionally4, NI3 was prepared by simply dropping a small amount (up to 1g) of I2 (s) into 0.880 S.G. ammonia, leaving for five minutes and filtering off as and when required in order to demonstrate its explosive capabilities. This procedure yields only small amounts of the triiodide as only a small surface area of iodine is in contact with the ammonia.
4 Hambly, G.F., Peters, R. Explosion of Nitrogen Triiodide : A safer and cleaner demonstration. J. Chem Ed. 943, 70 (11), 1993
My method was to grind up the preweighed iodine in a mortar and pestle, then to place the iodine into the ammonia. This yields a far higher amount of NI3 for the amount of iodine used.
3.3 Agar - chemical constitution.
Agar powder is a yellow crystalline solid with a low solubility at room temperature (less than 0.5 g per 100 cm3 water) and a much higher solubility at elevated temperatures (for some lower strength agars, upto 7 g will dissolve in 100 cm3 water).
Agar is a mixture of polysaccharides, containing 1 - 5% sulphate ester groups and has 2 main components - agarose and agaropectin. Agarose is the major constituent (~ 70%) and is a linear polymer of equimolar proportions of 1,3 - linked D - galactose and 1,4 - linked 3,6 - anhydro - L - galactose units. Agaropectin (the other ~ 30%) has a more complex structure than agarose by having in addition to the components of agarose, D - glucuronic acid, pyruvic acid and a far higher proportion of sulphate ester groups5.
5 Alger, M.S.M., Polymer Science Dictionary. p. 10. Elsevier Applied Science
This could be the reason for the low solubility at room temperature, the sulphate ester compounds do not lend themselves well to any reaction with water, but at the elevated temperatures, the C-N-S matrix can break. When cooled, the water is trapped in the gel. This can be born out by leaving an agar gel open to the atmosphere for a day. The gel will dry and shrivel.
