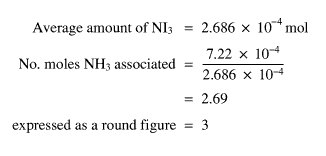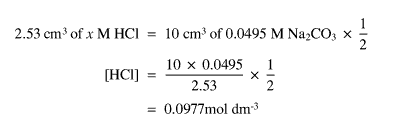
Determination of ammonia in the adduct. Method 1.11
1. Standardisation of HCl(aq)
1.3129g Na2CO3 anh. (AR grade, dried at 150°C for 5 hours) was dissolved in distilled water and made up to a final volume of 250 cm3.
This gave a solution of concentration 0.0495 mol dm-3. Sodium carbonate was used as it a primary standard.
8.7 cm3 of conc. hydrochloric acid was pipetted out and diluted to a final volume of 1 dm. This gave a concentration of 0.1 mol dm-3.
A 10 cm3 aliquot of the standard carbonate was pipetted into a 100 cm3 conical flask. Three drops of indicator (phenolphthalein) was added. The solution turned pink. The acid was poured into a 50 cm3 burette. The solutions were titrated in triplicate and the end - point determined by the solution going clear. A pH probe and meter were also used to ensure the end - point was found accurately.
| Solution number | Start titre (cm3) | End titre (cm3) | Titre (cm3) |
| 1 | 0.2 | 2.7 | 2.5 |
| 2 | 3 | 5.6 | 2.6 |
| 3 | 6 | 8.5 | 2.5 |
The three titres gave an average titre of 2.53 cm3.
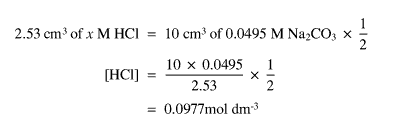
2. Concentration of ammonia evolved.
This test was carried out three times. The concentration of NI3 was determined using the mass of I2 used and assuming a 100% conversion of I2 to NI3.
| Sample number | Mass of I2 used | No. moles I2 | No moles NI3 produced |
| 1 | 0.3017 | 1.1887 x 10-3 | 2.5478 x 10-4 |
| 2 | 0.3651 | 1.4385 x 10-3 | 3.0832 x 10-4 |
| 3 | 0.2874 | 1.1323 x 10-3 | 2.4270 x 10-4 |
10 cm3 of the standardised HClaq was used for each test. At the end of each test, the acid was titrated against the standard sodium carbonate(aq) and the concentration of ammonia determined.
| Sample no. | Start titre (cm3) | End titre (cm3) | Titre (cm3) |
| 1 | 0.52 | 3.09 | 2.57 |
| 2 | 3.2 | 5.78 | 2.58 |
| 3 | 6 | 8.58 | 2.58 |
The average titre was 2.58 cm3. This meant that the concentration of the acid after the experiment was 0.0255 mol dm-3. The difference being 0.0733 mol dm-3 in HCl.
Taking the literature1 giving the formulae of the adduct as NI3.NH3 or NI3.3NH3, a relationship of one mole of NI3 to either one or three moles of NH3 was expected.
If the amount of NI3 is known, then the amount of the NH3 can be calculated thus
If there is one mole of NI3, there will be a corresponding x number of moles of NH3. Therefore, if there is y moles of NI3, there will be xy moles of NH3.
If the amount of NI3 is 5 x 10-4 moles and the ratio is 1 mole NI3 to 3 moles NH3, then 3 ´ (5 x 10-4) = true number of moles NH3 which will be in the adduct.
From my results, 7.22 x 10-4 moles NH3 was evolved from the adduct. If the number of moles NH3 evolved is divided by the average amount of NI3 the ratio is found